
Teaching Physics with the Physics Suite
Home | Action Research Kit| Sample Problems | Resources | Product Information
Problems Sorted by Type | Problems Sorted by Subject | Problems Sorted by Chapter in UP
 |
Teaching Physics with the Physics Suite
Home | Action Research Kit| Sample Problems | Resources | Product Information |
Problems Sorted by Type | Problems Sorted by Subject | Problems Sorted by Chapter in UP |
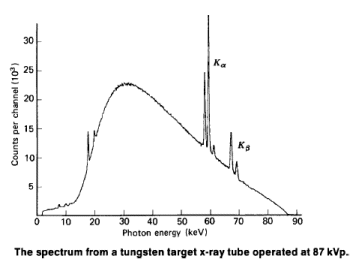
When high energy electrons are incident on a tungsten target, a broad spectrum of x-rays are given off. Most of the x-ray photons are spread over a range of energies, but there are a large number of x-rays with a specific energy -about 60 keV. This sharp x-ray line arises because:
|
|
Not finding what you wanted? Check the Site Map for more information.
Page last modified October 31, 2002: MP08