
Teaching Physics with the Physics Suite
Home | Action Research Kit| Sample Problems | Resources | Product Information
Problems Sorted by Type | Problems Sorted by Subject | Problems Sorted by Chapter in UP
 |
Teaching Physics with the Physics Suite
Home | Action Research Kit| Sample Problems | Resources | Product Information |
Problems Sorted by Type | Problems Sorted by Subject | Problems Sorted by Chapter in UP |
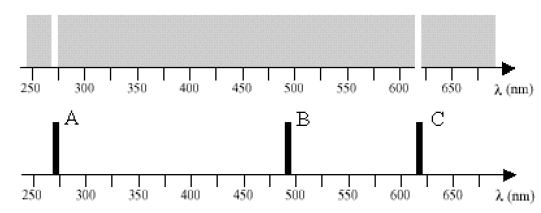
Observations of an interstellar gas give an absorption spectrum, a part of which is shown in the upper part of the figure below. The gas is cold, such that when not excited, all electrons are in the ground state. Astronomers suspect the gas to be a type of helium, but they aren't sure. When they excite a sample of helium in a spectrometer in the laboratory, they observe an emission spectrum, part of which is shown in the lower part of the figure below.
Not finding what you wanted? Check the Site Map for more information.
Page last modified October 31, 2002: MP10