
Teaching Physics with the Physics Suite
Edward F. Redish
Home | Action Research Kit| Sample Problems | Resources | Product Information
Problems Sorted by Type | Problems Sorted by Subject | Problems Sorted by Chapter in UP
 |
Teaching Physics with the Physics Suite Edward F. Redish Home | Action Research Kit| Sample Problems | Resources | Product Information |
Problems Sorted by Type | Problems Sorted by Subject | Problems Sorted by Chapter in UP |
| Our knowledge of electric forces between charges gives us the opportunity to begin to understand the forces that hold matter together. As a simple example, consider a crystal of salt - Sodium Chloride (NaCl). Each molecule of salt separates into a positive sodium ion (Na+) and a negative chloride ion (Cl-). In a crystal of salt, these ions are arrayed alternately in a three-dimensional cubical array as shown in the figure at the right. We ask whether or not the electrostatic forces between the ions might be able to hold it together. | 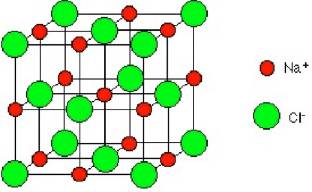 |
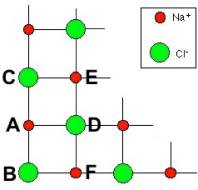 |
To simplify our analysis for the purpose of this problem, consider a single plane of this 3D structure. Let's look at an edge of one of these planes, shown in the figure at the left. We will analyze whether in the indicated arrangement, the net electrostatic force on a charge on the edge of the crystal is into the crystal (holding the crystal together) or out of the crystal (tending to break the crystal up). (a) First consider the force between an ion on the edge and one of its neighbors on the edge, for example, the sodium ion labeled A and the chloride ion labeled B. If the charge on the ions each have a magnitude e = 1.6 x 10-19 C and the lattice spacing is d (the distance between neighboring ions along the indicated lines), write an expression for the magnitude of the force between neighboring ions, F0. Express your answer in terms of the symbols e, d, and whatever constants you require |
(b) Now consider the attraction on the sodium ion at A from its three nearest chloride neighbors, labeled B, C, and D. Find the magnitude and direction of the net force on A due to these three charges. Express your answer in terms of F0 for ease of interpretation.
(c) Then, consider the repulsion on the sodium ion at A from its two nearest sodium neighbors, labeled E and F. Find the magnitude and direction of the net force on A due to these two charges. Again, express your answer in terms of F0 for ease of interpretation.
(d) We only considered nearest neighbors. Why do you think we did this? Do you think this is a reasonable approximation? Give a brief justification for your answer.
Page last modified 2. April, 2009:E56