- (a)
- A steel rod has a length of exactly 20 cm at
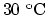 . How much longer is it at
. How much longer is it at 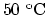 ?
The coefficient of linear expansion of steel is
?
The coefficient of linear expansion of steel is
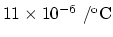 .
(5 points)
.
(5 points)
- (b)
- The coefficient of linear expansion of a certain solid
substance is
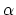 . Suppose that you made a square plate out of
the substance
of size
. Suppose that you made a square plate out of
the substance
of size 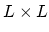 . Then what is the coefficient of area
expansion (i.e.,
. Then what is the coefficient of area
expansion (i.e.,
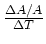 where
where 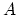 is the area)
of this plate? Prove your answer, neglecting the terms proportional to
is the area)
of this plate? Prove your answer, neglecting the terms proportional to
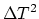 or higher order terms of
or higher order terms of 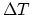 . (10 points)
. (10 points)
- (c)
- Normally, you can open a bottle of coke without spilling any of the liquid. But if you shake the bottle and immediately take the cap off (just to see if your cap is a winning cap), the coke will spew out everywhere even though you have not changed the temperature of the coke (from the act of shaking the bottle). Explain why this happens. (5 points)
- 2)
- Answer the following questions on specific heat and latent heat.
- (a)
- A metal of mass
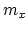 is heated to a temperature
is heated to a temperature 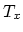 and
then dropped into a container containing a liquid of mass
and
then dropped into a container containing a liquid of mass 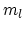 initially
at temperature
initially
at temperature 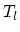 , where
, where 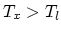 . If the final equilibrium temperature
is
. If the final equilibrium temperature
is 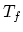 , find the specific heat of the metal. The specific heat of the
liquid is
, find the specific heat of the metal. The specific heat of the
liquid is 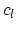 . (5 points)
. (5 points)
- (b)
- Silver has (i) specific heat of
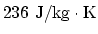 , (ii)
latent heat of fusion
, (ii)
latent heat of fusion
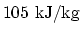 , and (iii) latent heat of
vapourisation
, and (iii) latent heat of
vapourisation
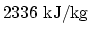 . The melting point of silver is at
1235 K and the boiling point at 2323 K. Given this information,
calculate the minimum amount of heat, in joules, required to
completely melt 130 g of silver initially at
. The melting point of silver is at
1235 K and the boiling point at 2323 K. Given this information,
calculate the minimum amount of heat, in joules, required to
completely melt 130 g of silver initially at 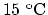 . Assume that the
specific heat does not change with temperature. (10 points)
. Assume that the
specific heat does not change with temperature. (10 points)
- (c)
- The internal energy of
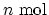 of
a certain gas can be expressed in terms of temperature as
of
a certain gas can be expressed in terms of temperature as
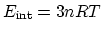 . What is the
. What is the 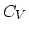 (constant-volume molar specific heat) of this gas? (5 points)
(constant-volume molar specific heat) of this gas? (5 points)
- 3)
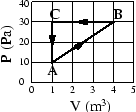
- An ideal monatomic gas within a chamber undergoes the processes shown
in the
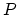 -
-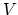 diagram of Fig. 1.
diagram of Fig. 1.
- (a)
- Calculate the net heat added to the system during one complete cycle. Give the correct sign of the heat. (5 points)
Calculate the work done by the gas and calculate the heat added to the gas during (b) process
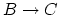 (5 points), (c) process
(5 points), (c) process
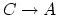 (5 points), and (d) process
(5 points), and (d) process
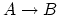 (5 points). Hint:
The internal energy of an ideal monatomic
gas is
(5 points). Hint:
The internal energy of an ideal monatomic
gas is
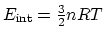 .
.
- 4)
- A vertical cylinder with a heavy piston contains air at 300
K. The initial pressure is 200 kPa, and the initial volume is
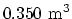 . Take the molar mass of air as
. Take the molar mass of air as
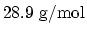 and assume
that
and assume
that 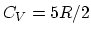 .
.
- (a)
- Find the specific heat of air at constant
volume in units of
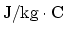 . (5 points)
. (5 points)
- (b)
- What can you say about the number of degrees of freedom of each molecule of the air? Estimate the r.m.s. speed of the air molecules in the initial state and compare it to that of an ideal monatomic gas at the same temperature. Are they equal or not? (10 points)
- (c)
- Assume the condition of the initial state and that the heavy piston is free to move. Find the energy input required to raise temperature to 700 K. (5 points)
- 5)
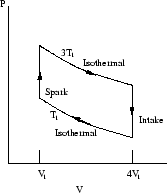
- Figure 2 shows
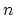 mol of an ideal monatomic
gas being taken through a reversible cycle that consists of two isothermal
processes at temperatures
mol of an ideal monatomic
gas being taken through a reversible cycle that consists of two isothermal
processes at temperatures 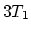 and
and 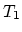 and two constant-volume
processes with volumes
and two constant-volume
processes with volumes 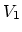 and
and 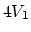 .
.
- (a)
- What is the efficiency of the cycle? Give the derivation.
(15 points)
- (b)
- What is the total entropy change in one cycle? In realistic engines with irreversible processes, what can you say about the total entropy change in the whole system (engine + surroundings)? (5 points)
Next: Solutions Up: Mid-term (PHYS262, Summer 2001) Previous: Mid-term (PHYS262, Summer 2001) Hyok-Jon Kwon
2001-08-10